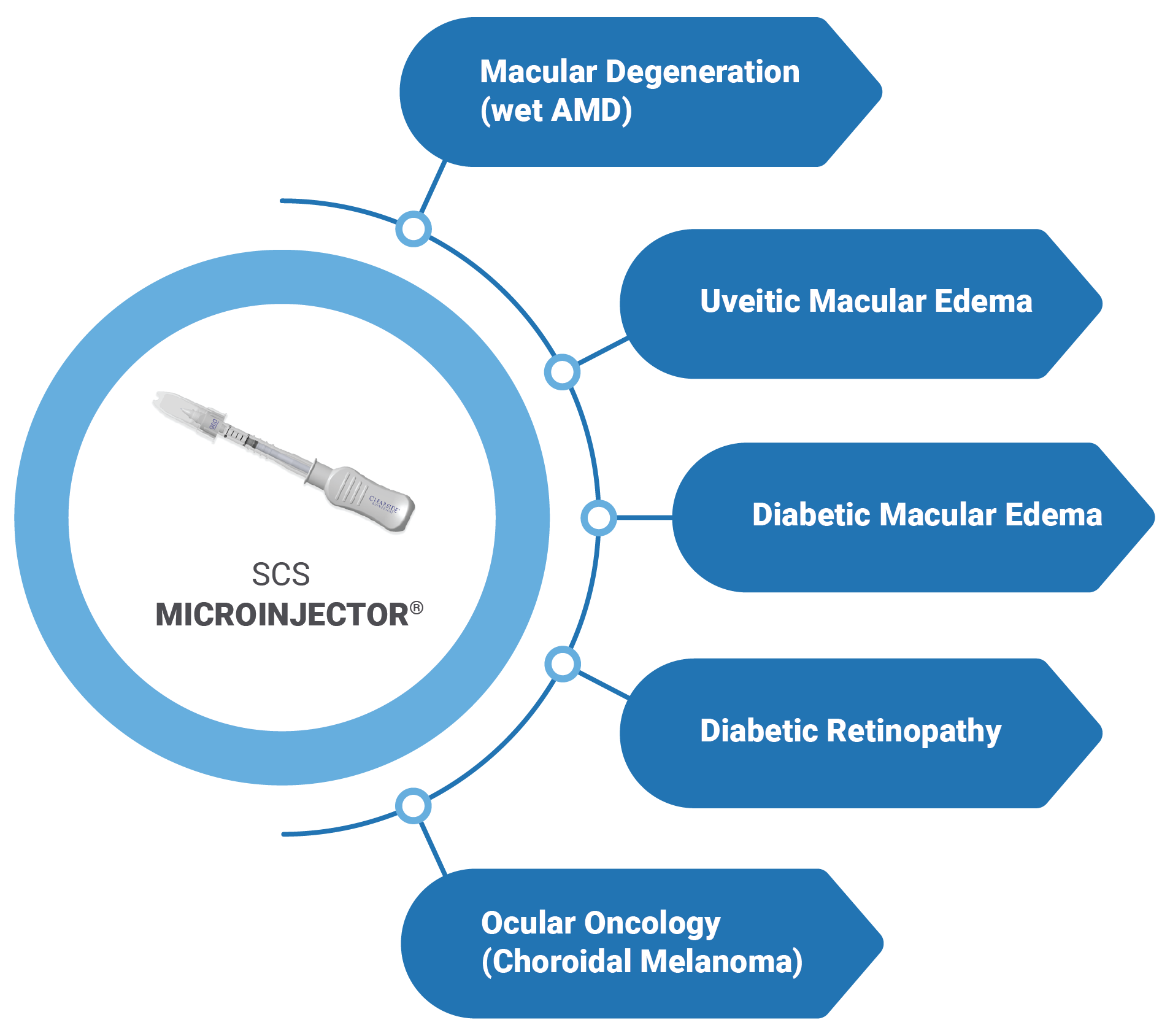
Diversified Portfolio Targeting Multiple Retinal Disorders
Together with our partners, we are seeking new treatments for multiple blinding eye diseases affecting millions of people worldwide

INTELLECTUAL PROPERTY
Our extensive patent portfolio provides us with exclusive rights to commercialize pharmacological agents for treatment of eye diseases via suprachoroidal injection. Our drug candidates, SCS Microinjector and method of drug administration into the SCS are protected by multiple U.S., European and international patents broadly directed to the use of the device, administration of any drug into the SCS by injection, as well as specific product candidates.