Tyrosine kinase inhibitor (TKI) with potential to treat VEGF-driven disorders such as wet AMD, diabetic macular edema and diabetic retinopathy
High potency and pan-VEGF attributes of TKI axitinib
Targeted delivery via SCS Microinjector®
Enhanced durability via small molecule suspension
Suprachoroidal Injection of CLS-AX Provides Greater Targeted Delivery
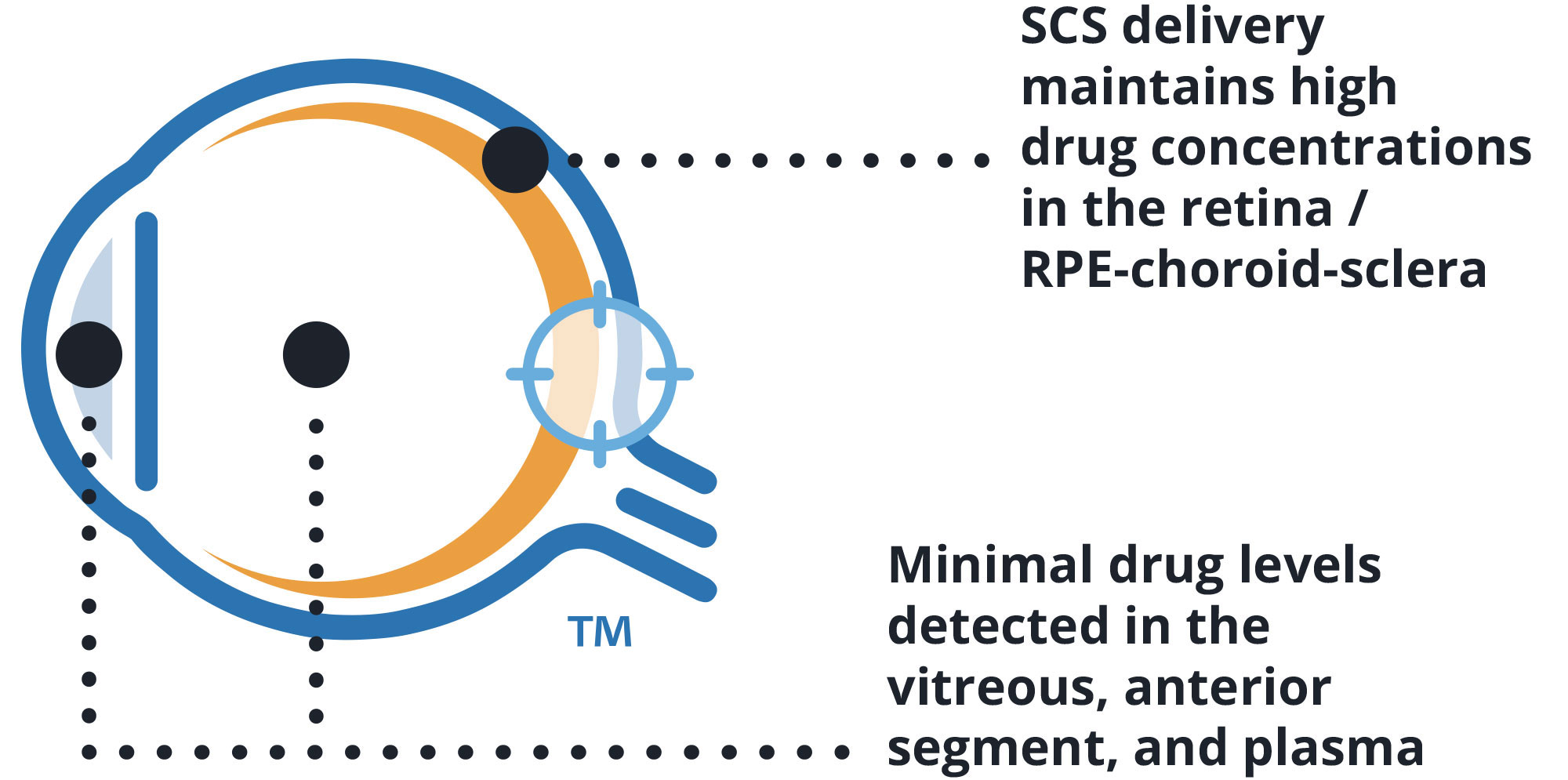
Abbreviations: SCS: Suprachoroidal Space | RPE: Retinal pigment epithelium

CLS-AX Phase 2b Clinical Trial in Wet AMD
Additional information on the ODYSSEY Phase 2b trial can be found on ClinicalTrials.gov NCT05891548.
References:
Medscape: F Ryan Prall, MD, et al Assistant Professor of Ophthalmology, Indiana University School of Medicine | Pennington, Katie L and DeAngelis, Margaret M Eye and Vision, Epidemiology of age-related macular degeneration (AMD): associations with cardiovascular disease phenotypes and lipid factors, Dec 22, 2016. | Cabral T et al. Bevacizumab Injection in Patients with Neovascular Age-Related Macular Degeneration Increases Angiogenic Biomarkers. Ophthalmol Retina. 2018 January; 2(1): 31–37. doi:10.1016/j.oret.2017.04.004. | Lieu et al. The Association of Alternate VEGF Ligands with Resistance to Anti-VEGF Therapy in Metastatic Colorectal Cancer. PLoS ONE 8(10): e77117. | Ciulla TA et al. Ophthalmology Retina. 2019 May 25. pii: S2468-6530(19)30280-5. | Viral S. Kansara, Leroy W. Muya, Thomas A. Ciulla; Evaluation of Long-Lasting Potential of Suprachoroidal Axitinib Suspension Via Ocular and Systemic Disposition in Rabbits. Trans. Vis. Sci. Tech. 2021;10(7):19.